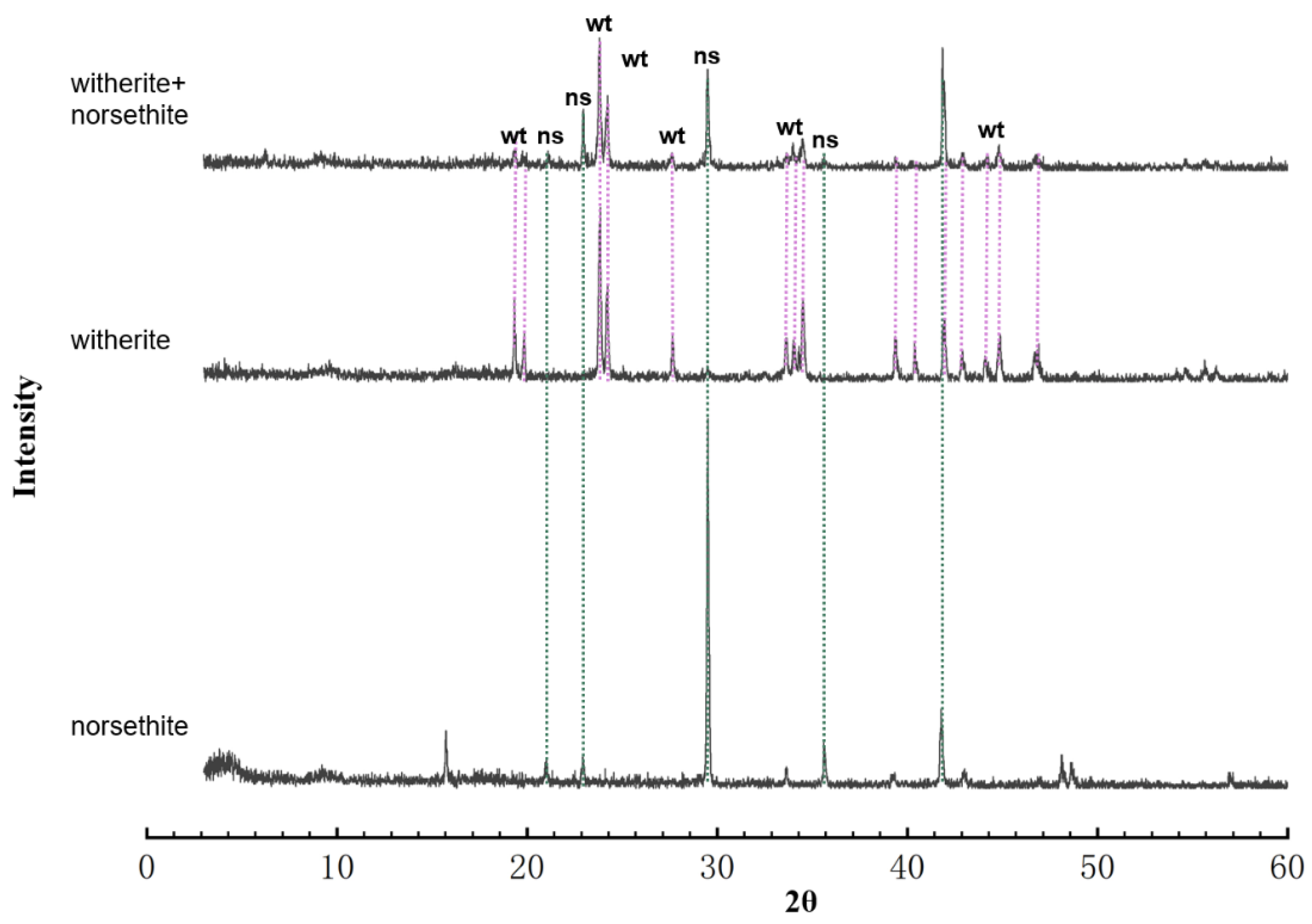
Precipitation of Ordered Dolomite via Simultaneous Dissolution of Calcite and Magnesite: New Experimental Insights into an Old Precipitation Enigma | Crystal Growth & Design

Factors controlling and influencing polymorphism, morphology and size of calcium carbonate synthesized through the carbonation route: A review - ScienceDirect

SE - Precipitation of dolomite from seawater on a Carnian coastal plain ( Dolomites, northern Italy): evidence from carbonate petrography and Sr isotopes

Minerals | Free Full-Text | The Effect of Stoichiometry, Mg-Ca Distribution, and Iron, Manganese, and Zinc Impurities on the Dolomite Order Degree: A Theoretical Study

Magnesium Ions Direct the Solid‐State Transformation of Amorphous Calcium Carbonate Thin Films to Aragonite, Magnesium‐Calcite, or Dolomite - Zhang - 2022 - Advanced Functional Materials - Wiley Online Library

Non‐classical crystallization of very high magnesium calcite and magnesite in the Coorong Lakes, Australia - Raudsepp - 2022 - Sedimentology - Wiley Online Library

Structure, genesis and resources efficiency of dolomite: New insights and remaining enigmas - ScienceDirect
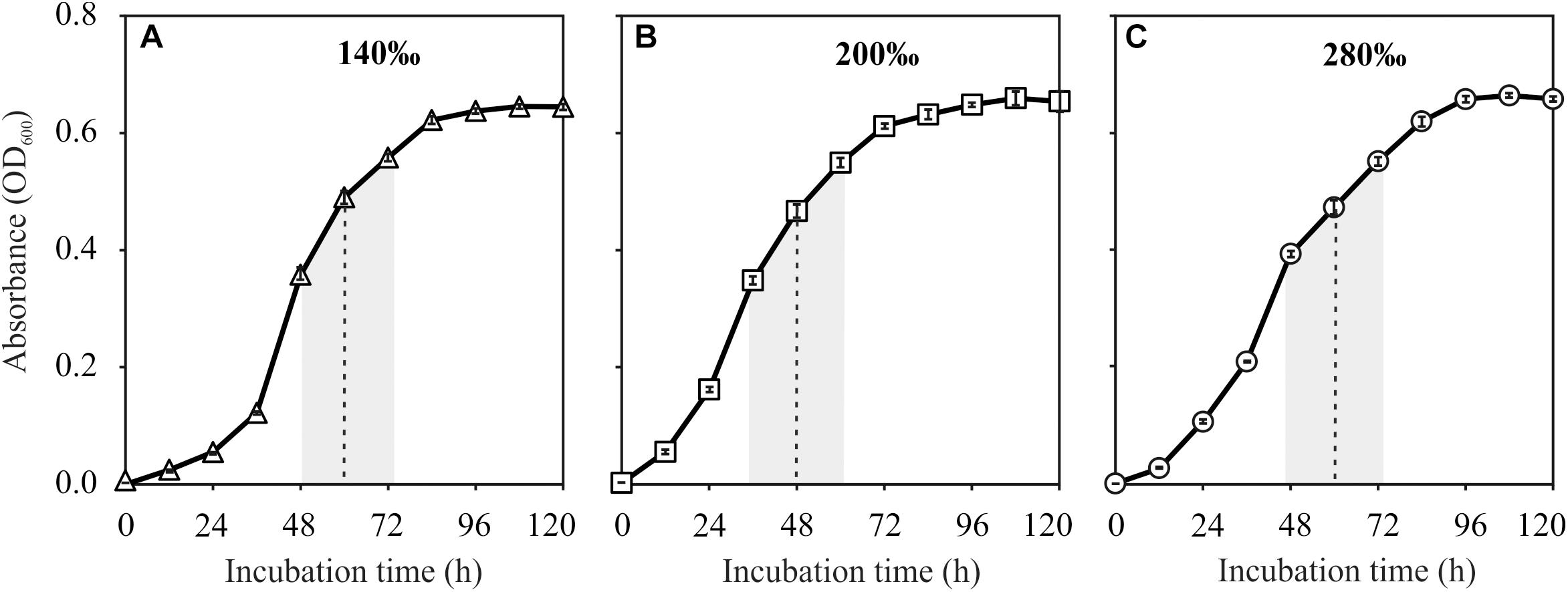
Frontiers | Halophilic Archaea Mediate the Formation of Proto-Dolomite in Solutions With Various Sulfate Concentrations and Salinities
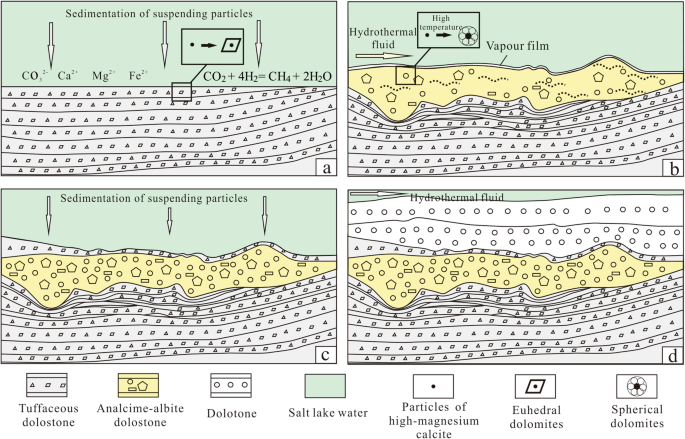
Hydrothermal-sedimentary dolomite — a case from the Middle Permian in eastern Junggar Basin, China | Journal of Palaeogeography | Full Text

Precipitation of Ordered Dolomite via Simultaneous Dissolution of Calcite and Magnesite: New Experimental Insights into an Old Precipitation Enigma | Crystal Growth & Design

Solubility product constants for natural dolomite (0–200 °C) through a groundwater-based approach using the USGS produced water database | American Journal of Science

Effects of early diagenesis on Mg isotopes in dolomite: The roles of Mn(IV)-reduction and recrystallization - ScienceDirect
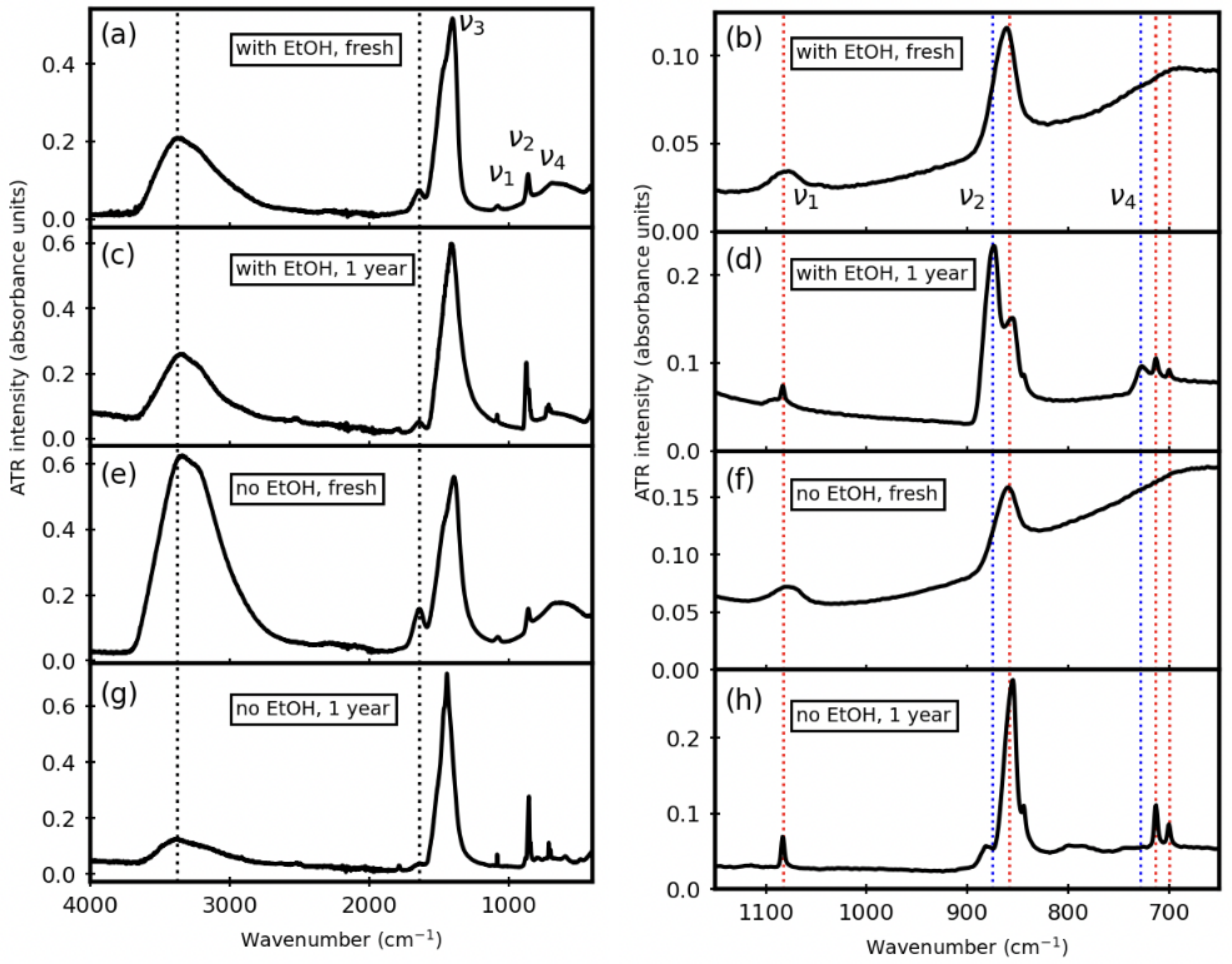
Minerals | Free Full-Text | Tracking Amorphous Calcium Carbonate Crystallization Products with Far-Infrared Spectroscopy

Precipitation of Ordered Dolomite via Simultaneous Dissolution of Calcite and Magnesite: New Experimental Insights into an Old Precipitation Enigma | Crystal Growth & Design