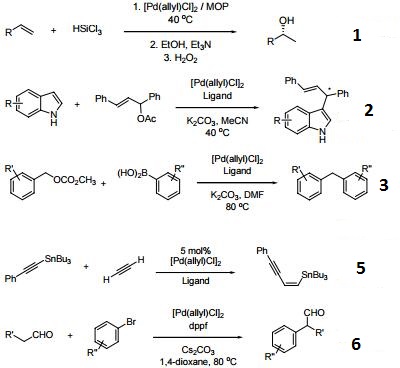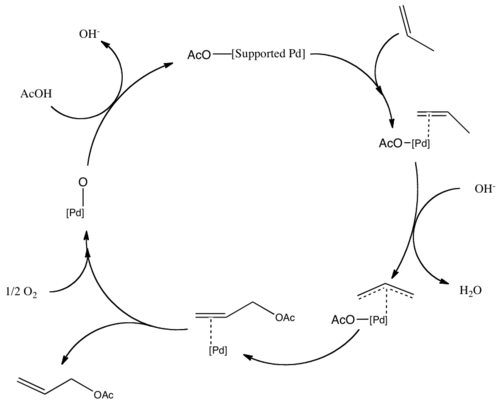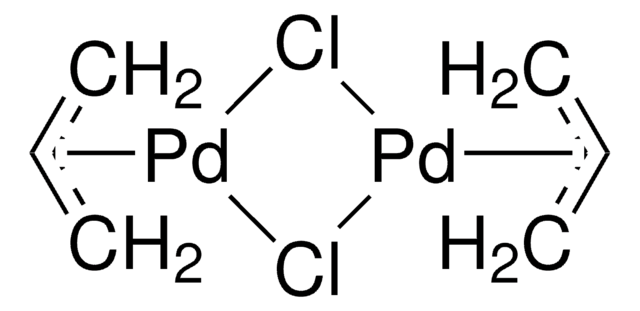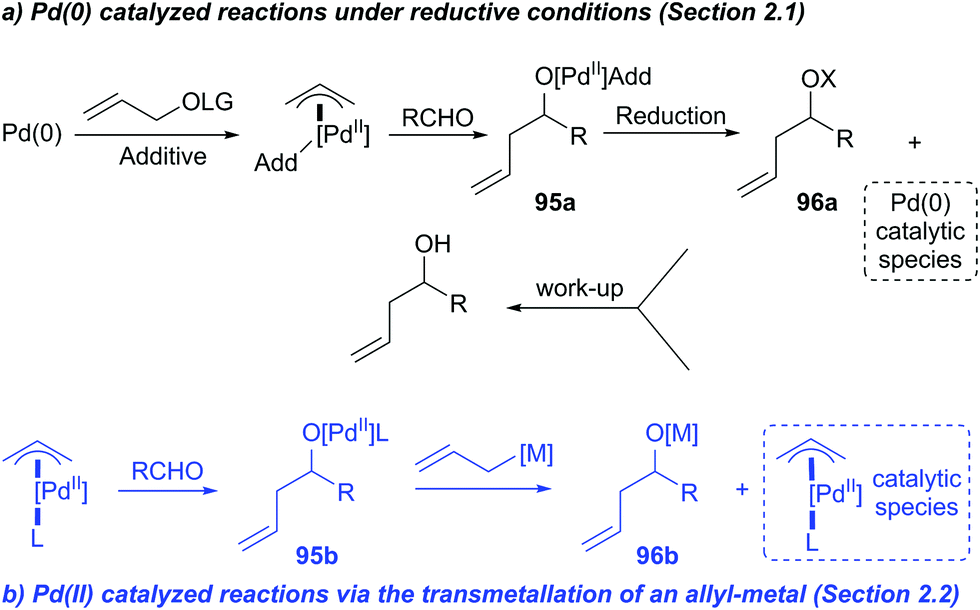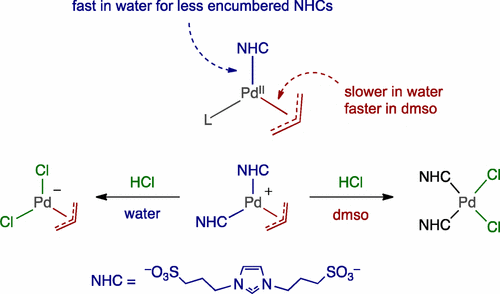
Aqueous-Phase Chemistry of η3-Allylpalladium(II) Complexes with Sulfonated N-Heterocyclic Carbene Ligands: Solvent Effects in the Protolysis of Pd–C Bonds and Suzuki–Miyaura Reactions - Organometallics - X-MOL

Synthesis and characterization of (π-allyl)palladium(II) complexes containing dialkylbiaryl phosphine ligands - ScienceDirect
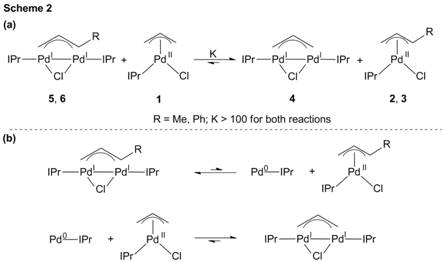
Report: Palladium(I) and Nickel(I) Bridging Allyl Dimers for the Catalytic Functionalization of CO2 (58th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)
A Mechanistic Study of Direct Activation of Allylic Alcohols in Palladium Catalyzed Amination Reactions

Stereochemistry of the palladium-catalyzed allylic substitution: the syn-anti dichotomy in the formation of (π-allyl)palladium complexes and their equilibration - ScienceDirect
![PDF] Confirming the existence of π-allyl-palladium intermediates during the reaction of meta photocycloadducts with palladium(II) compounds. | Semantic Scholar PDF] Confirming the existence of π-allyl-palladium intermediates during the reaction of meta photocycloadducts with palladium(II) compounds. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e3170611736068557037b936b6641e36bc061f7e/4-Figure2-1.png)
PDF] Confirming the existence of π-allyl-palladium intermediates during the reaction of meta photocycloadducts with palladium(II) compounds. | Semantic Scholar
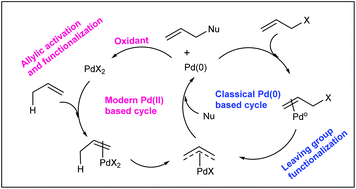
Catalytic allylic functionalization via π-allyl palladium chemistry - Organic & Biomolecular Chemistry (RSC Publishing)

Steric Effects in Enantioselective Allylic Alkylation Catalysed by Cationic(η3‐Allyl)palladium Complexes Bearing Chiral Pyridine‐Aziridine Ligands - Ferioli - 2005 - European Journal of Organic Chemistry - Wiley Online Library

Palladium/N-heterocyclic carbene catalysed regio and diastereoselective reaction of ketones with allyl reagents via inner-sphere mechanism | Nature Communications

Palladium‐Catalyzed Electrophilic Allylation Reactions via Bis(allyl) palladium Complexes and Related Intermediates - Szabó - 2004 - Chemistry – A European Journal - Wiley Online Library