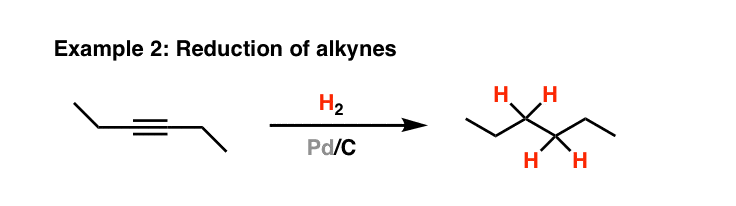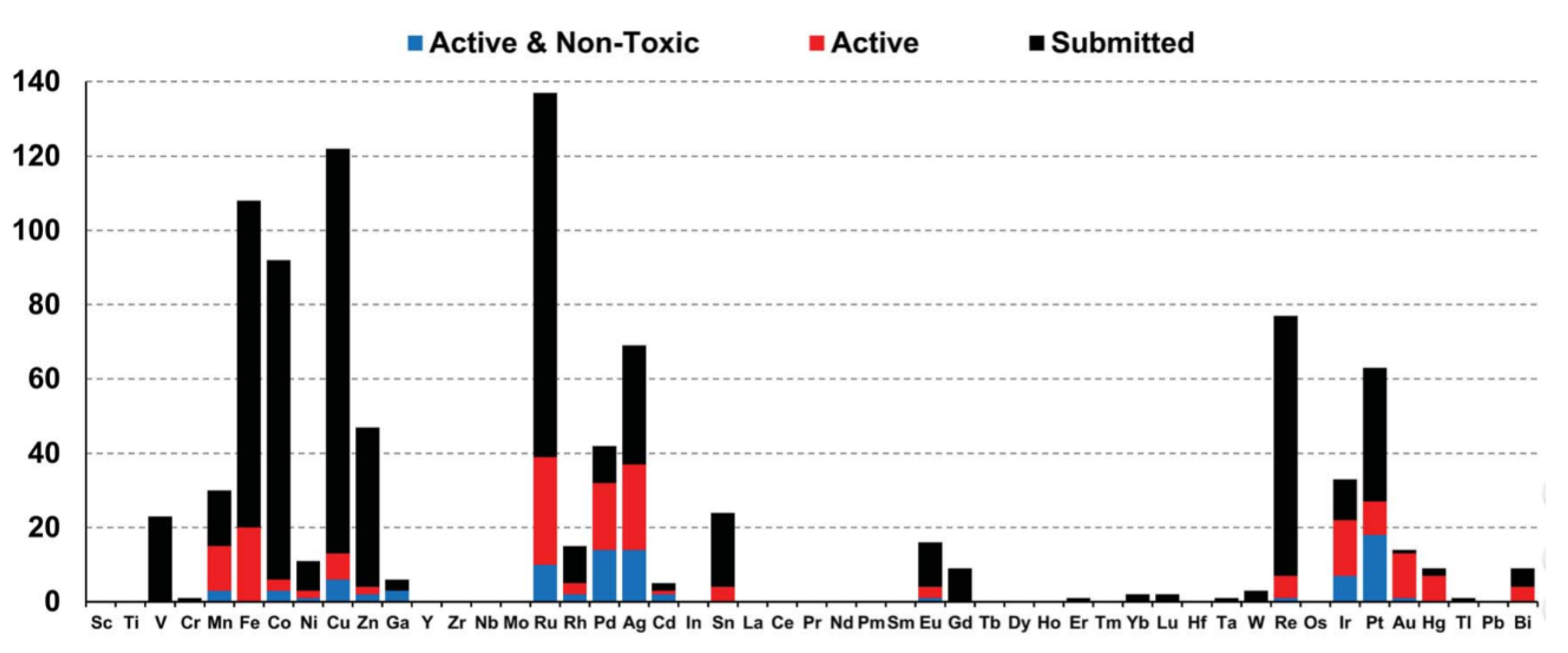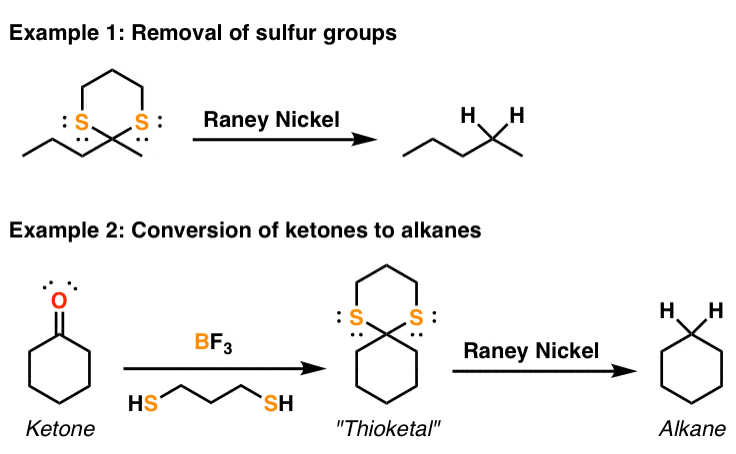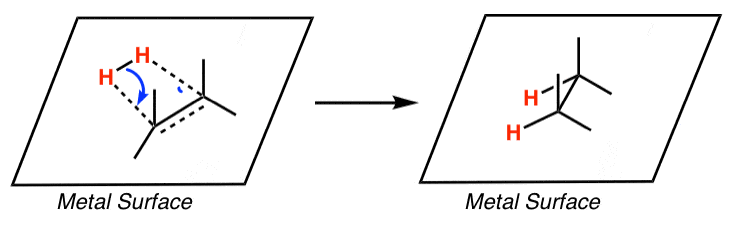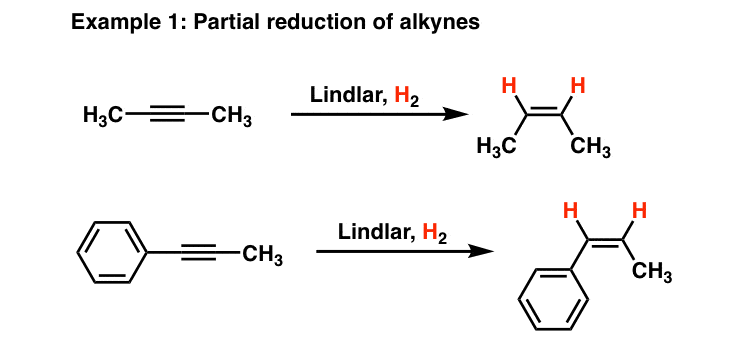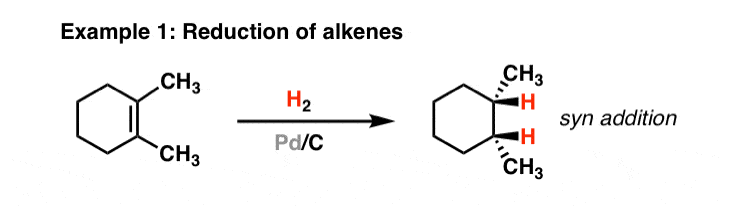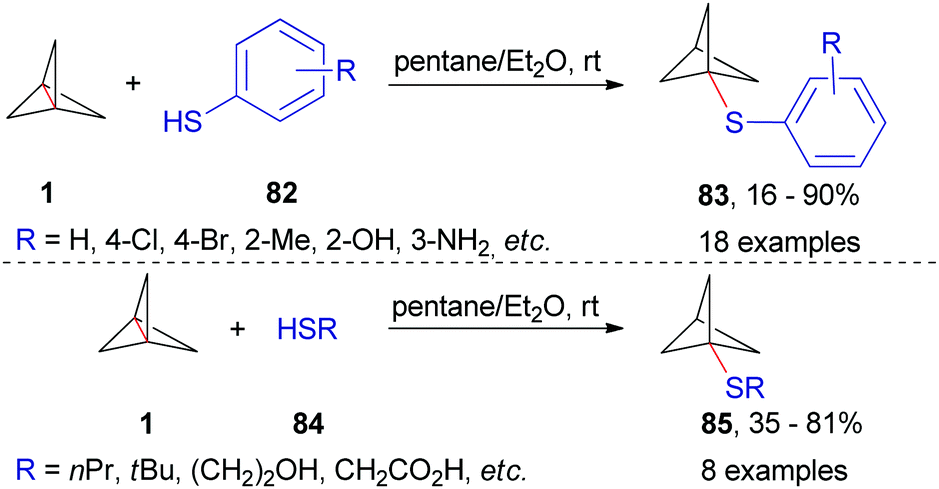
Strain release – an old tool for new transformations - Chemical Communications (RSC Publishing) DOI:10.1039/D0CC01771J

Selection and synthesis of amphiphilic imidazolium chlorides 1–8 for... | Download Scientific Diagram
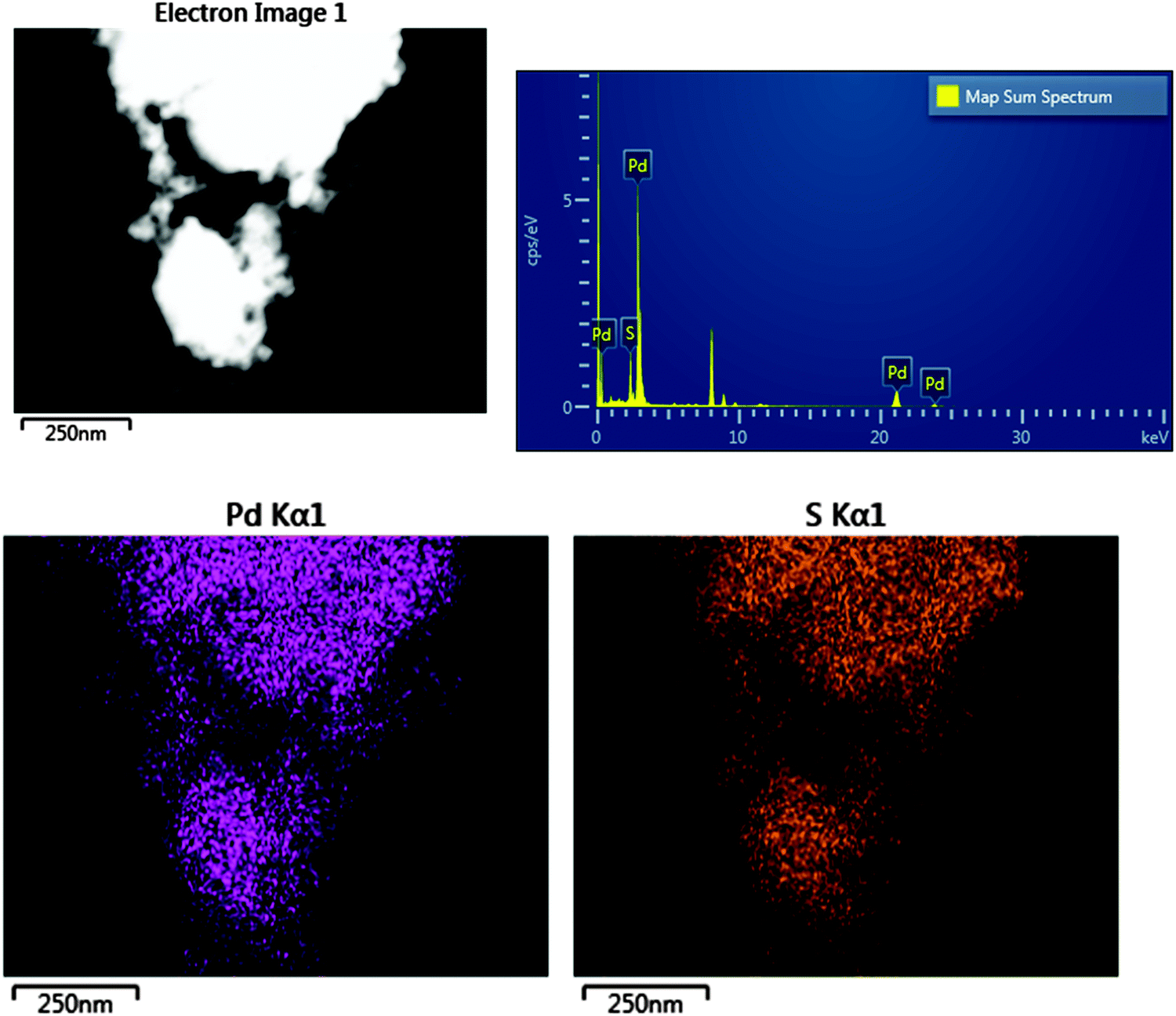
The adsorbed state of a thiol on palladium nanoparticles - Physical Chemistry Chemical Physics (RSC Publishing)

Why Does Industry Not Use Immobilized Transition Metal Complexes as Catalysts? - Hübner - 2016 - Advanced Synthesis & Catalysis - Wiley Online Library
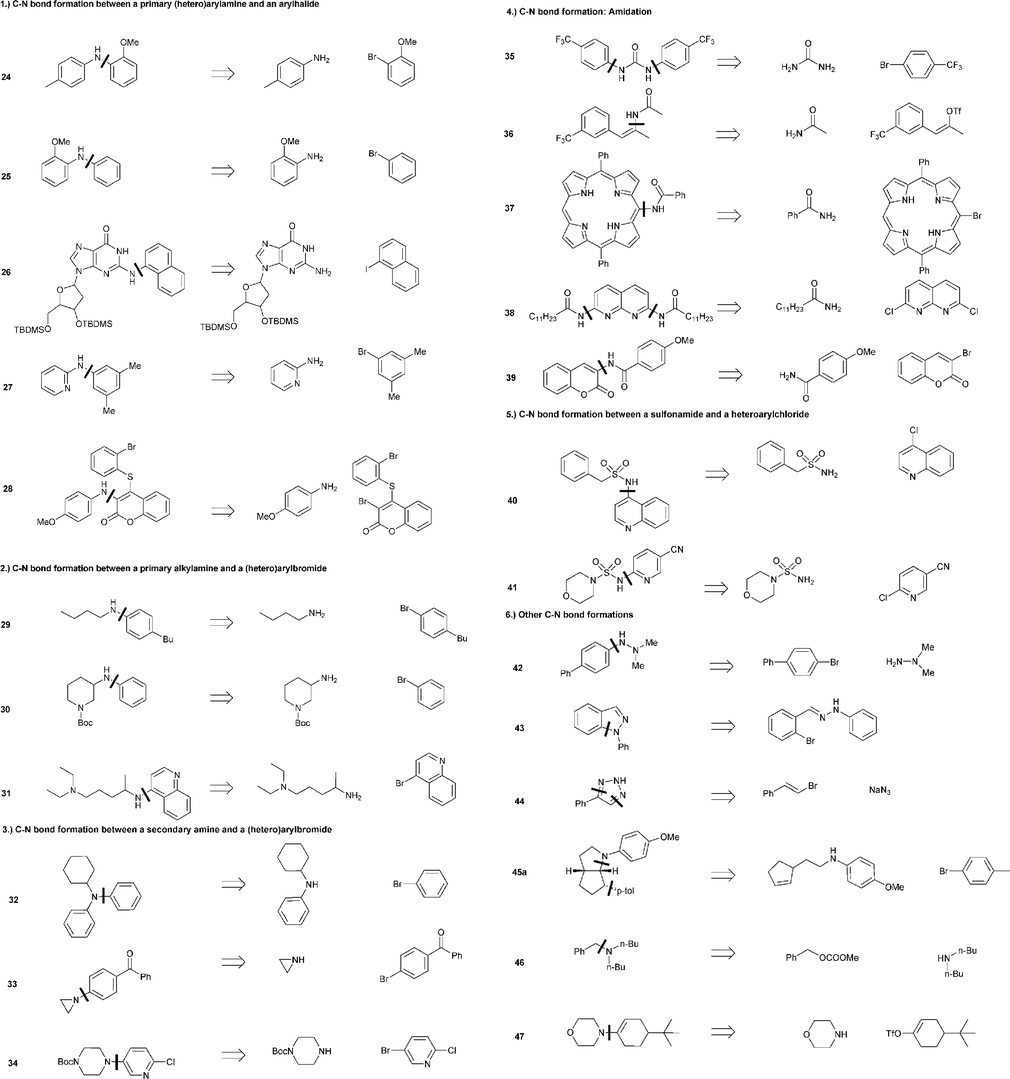
Bite angle effects of diphosphines in C–C and C–X bond forming cross coupling reactions - Chemical Society Reviews (RSC Publishing)
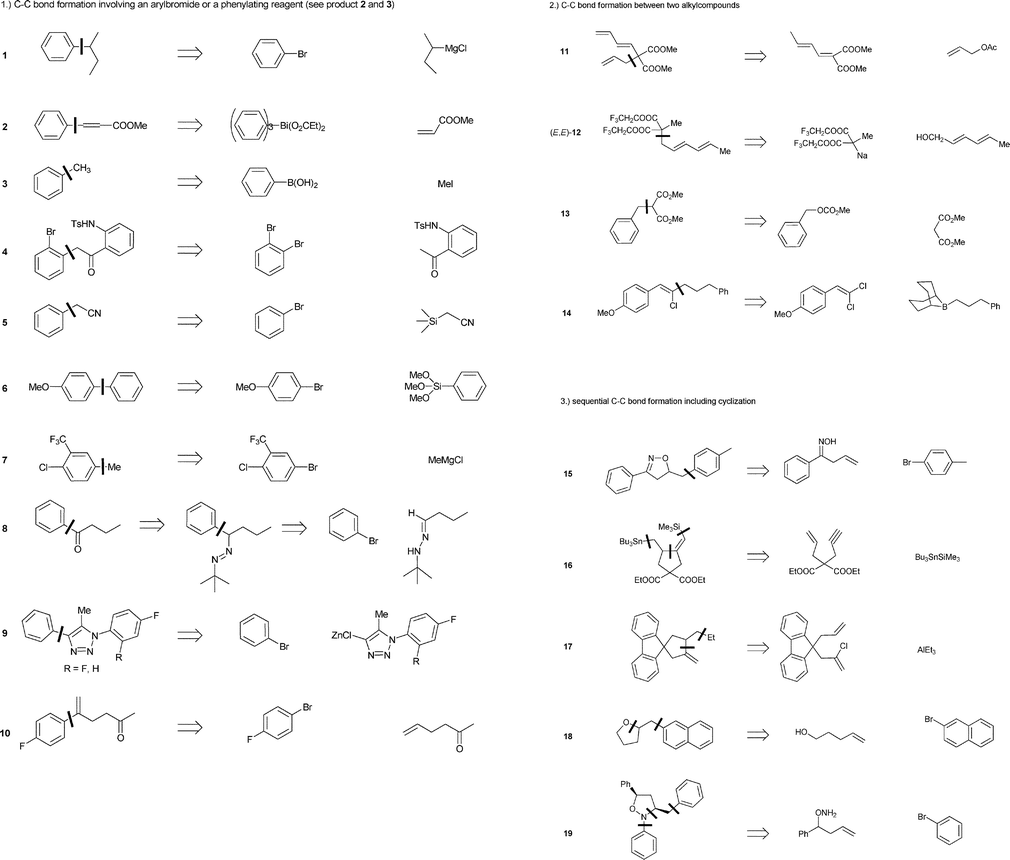
Bite angle effects of diphosphines in C–C and C–X bond forming cross coupling reactions - Chemical Society Reviews (RSC Publishing)

Why Does Industry Not Use Immobilized Transition Metal Complexes as Catalysts? - Hübner - 2016 - Advanced Synthesis & Catalysis - Wiley Online Library
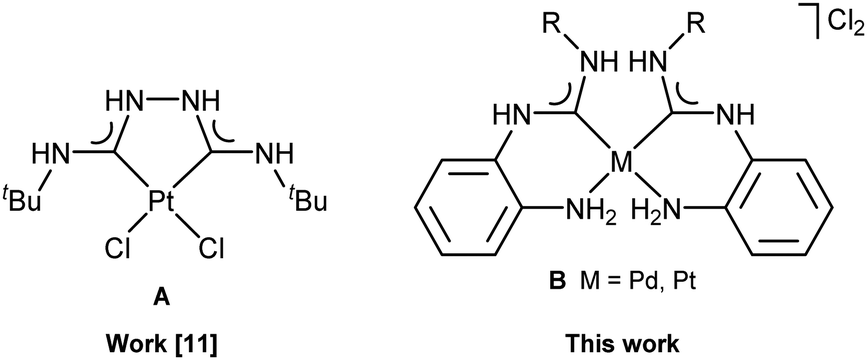
Water soluble palladium(ii) and platinum(ii) acyclic diaminocarbene complexes: solution behavior, DNA binding, and antiproliferative activity - New Journal of Chemistry (RSC Publishing)
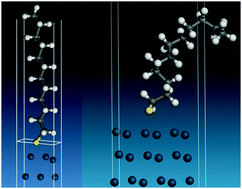
The adsorbed state of a thiol on palladium nanoparticles - Physical Chemistry Chemical Physics (RSC Publishing)

Metal assisted peptide bond hydrolysis: Chemistry, biotechnology and toxicological implications - ScienceDirect

a) The epoxybutene hydrogenation pathway; (b) the formation rate and... | Download Scientific Diagram
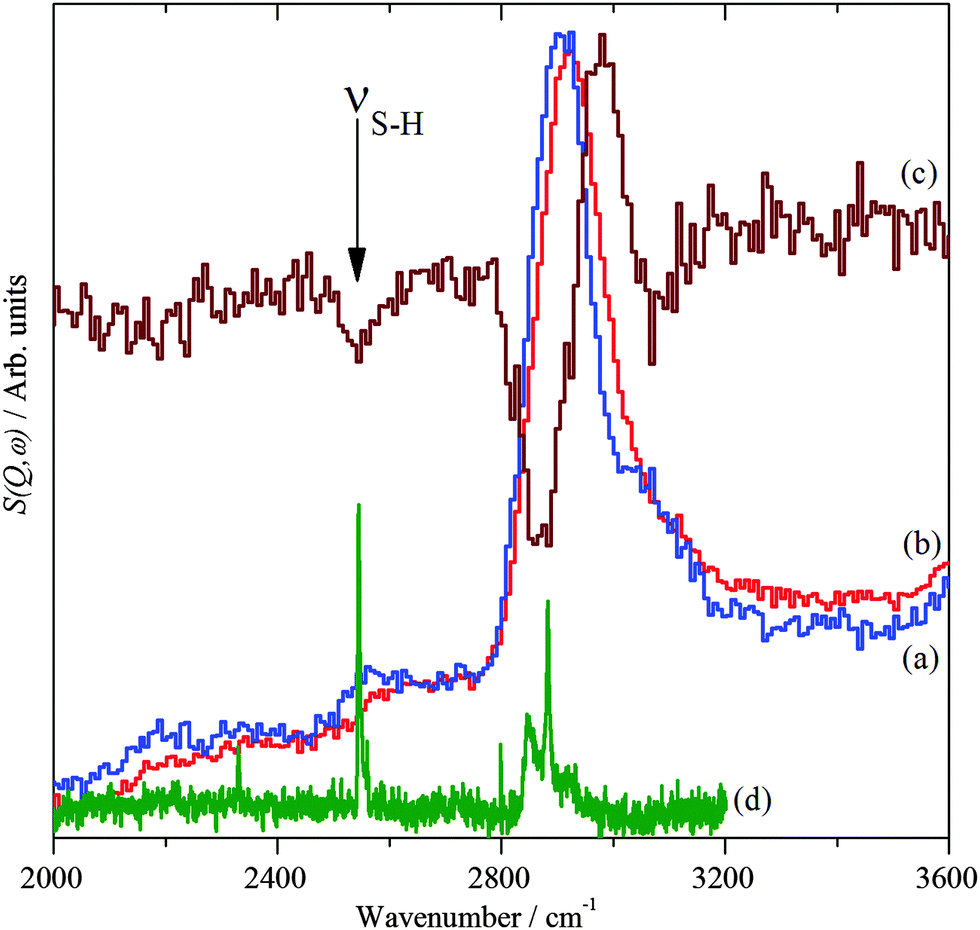
The adsorbed state of a thiol on palladium nanoparticles - Physical Chemistry Chemical Physics (RSC Publishing)
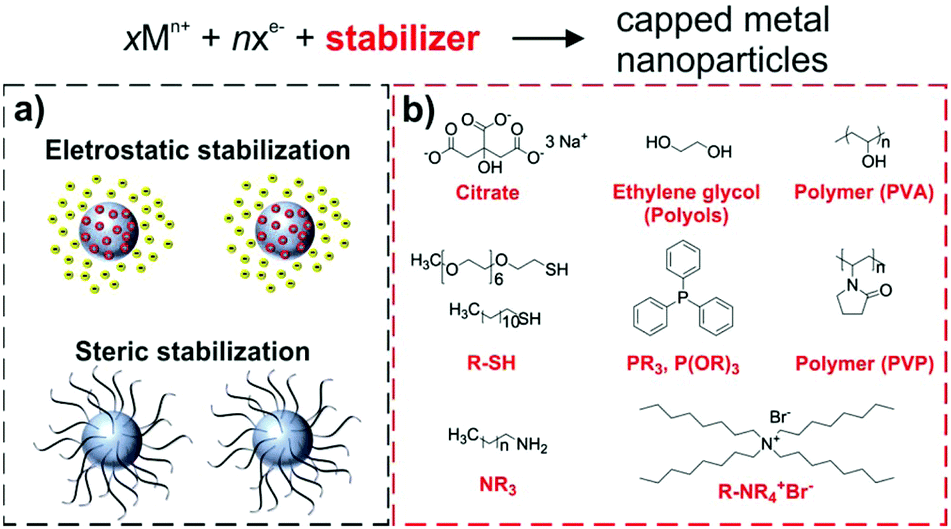
The role and fate of capping ligands in colloidally prepared metal nanoparticle catalysts - Dalton Transactions (RSC Publishing)


![Selected bond lengths of [Pd(BHEP)Cl 2 ]. | Download Table Selected bond lengths of [Pd(BHEP)Cl 2 ]. | Download Table](https://www.researchgate.net/profile/Mahmoud_Mabrouk11/publication/287806625/figure/tbl2/AS:614201169022979@1523448378198/Selected-bond-angles-of-PdBHEPCl-2_Q320.jpg)