Air-stable imidazole-imine palladium complexes for Suzuki–Miyaura coupling: Toward an efficient, green synthesis of biaryl compounds - ScienceDirect
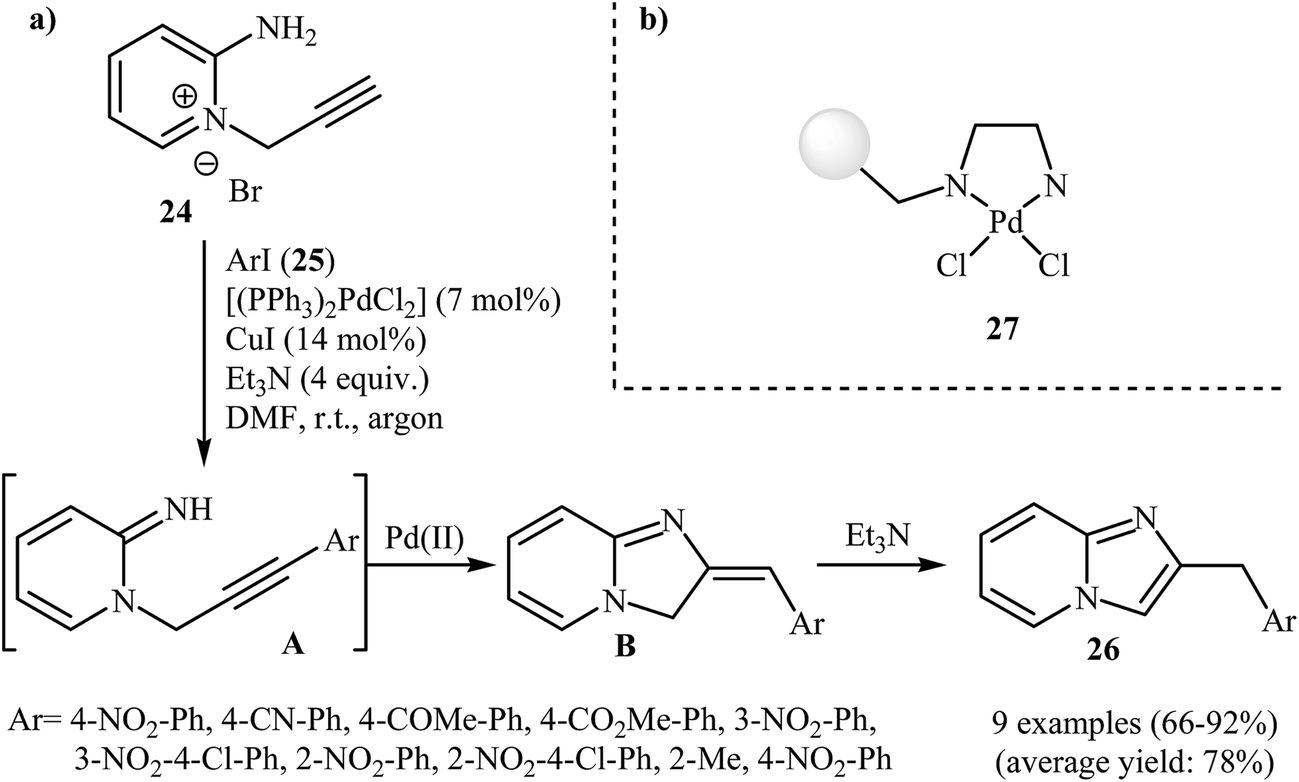
New protocols to access imidazoles and their ring fused analogues: synthesis from N -propargylamines - RSC Advances (RSC Publishing) DOI:10.1039/C6RA25816F
![Synthesis, Structural Characterization, and Coordination Chemistry of (Trineopentylphosphine)palladium(aryl)bromide Dimer Complexes ([(Np3P)Pd(Ar)Br]2) - Inorg. Chem. - X-MOL Synthesis, Structural Characterization, and Coordination Chemistry of (Trineopentylphosphine)palladium(aryl)bromide Dimer Complexes ([(Np3P)Pd(Ar)Br]2) - Inorg. Chem. - X-MOL](https://xpic.x-mol.com/20190926%2F10.1021_acs.inorgchem.9b02164.jpg)
Synthesis, Structural Characterization, and Coordination Chemistry of (Trineopentylphosphine)palladium(aryl)bromide Dimer Complexes ([(Np3P)Pd(Ar)Br]2) - Inorg. Chem. - X-MOL
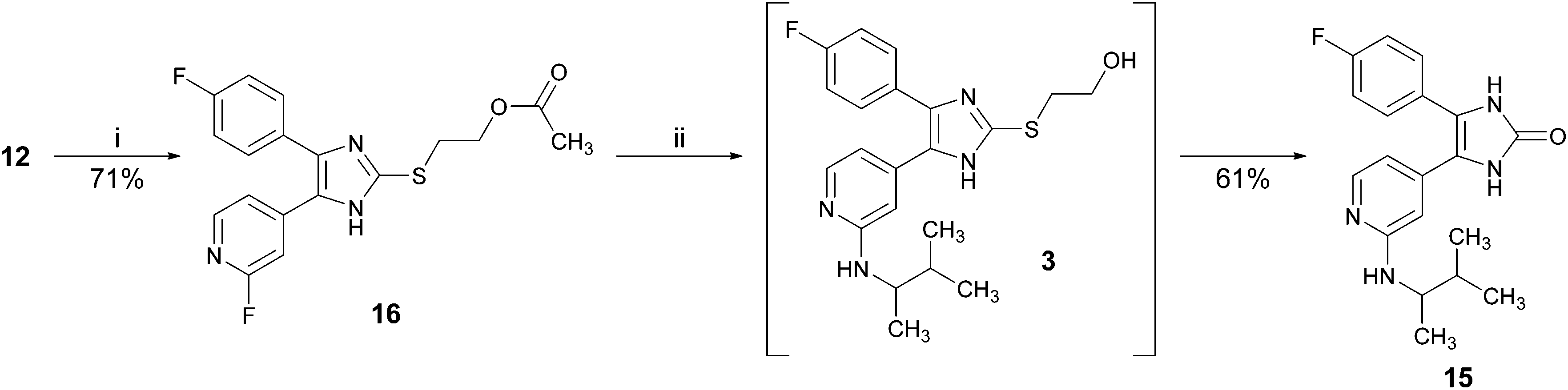
An optimized and versatile synthesis to pyridinylimidazole-type p38α mitogen activated protein kinase inhibitors - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB01505G

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

PDF) Efficient hydroarylation of terminal alkynes with sodium tetraphenylborate performed in water under mild conditions

Regiocontrolled Synthesis of 1,2‐Diaryl‐1H‐imidazoles by Palladium‐ and Copper‐Mediated Direct Coupling of 1‐Aryl‐1H‐imidazoles with Aryl Halides under Ligandless Conditions - Bellina - 2006 - European Journal of Organic Chemistry - Wiley Online Library

Are Imidazoles Versatile or Promiscuous in Reactions with Organophosphates? Insights from the Case of Parathion
![Direct arylation and Suzuki-Miyaura coupling of imidazo[1,2-a]pyridines catalyzed by (SIPr)Pd(allyl)Cl complex under microwave-irradiation | El Abbouchi | Mediterranean Journal of Chemistry Direct arylation and Suzuki-Miyaura coupling of imidazo[1,2-a]pyridines catalyzed by (SIPr)Pd(allyl)Cl complex under microwave-irradiation | El Abbouchi | Mediterranean Journal of Chemistry](http://medjchem.com/public/journals/1/cover_article_1124_en_US.png)
Direct arylation and Suzuki-Miyaura coupling of imidazo[1,2-a]pyridines catalyzed by (SIPr)Pd(allyl)Cl complex under microwave-irradiation | El Abbouchi | Mediterranean Journal of Chemistry

Imidazole-aryl coupling reaction via CH bond activation catalyzed by palladium supported on modified magnetic reduced graphene oxide in alkaline deep eutectic solvent - ScienceDirect

New platinum(II) and palladium(II) quinoline-imine-pyridine, quinoline-imine-thiazole and quinoline-imine-imidazole complexes by metal-assisted condensation reactions - ScienceDirect

Dimetallic Palladium‐NHC Complexes: Synthesis, Characterization, and Catalytic Application for Direct C−H Arylation Reaction of Heteroaromatics with Aryl Chlorides - Lee - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
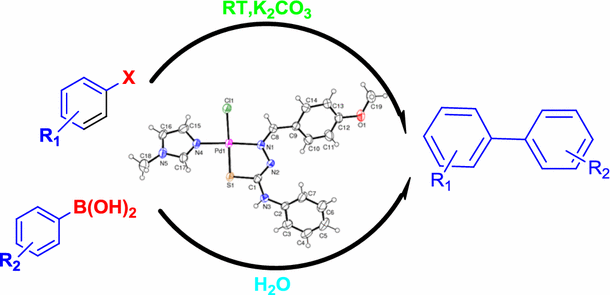
A thiosemicarbazone–palladium(II)–imidazole complex as an efficient pre-catalyst for Suzuki–Miyaura cross-coupling reactions at room temperature in aqueous media | SpringerLink

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
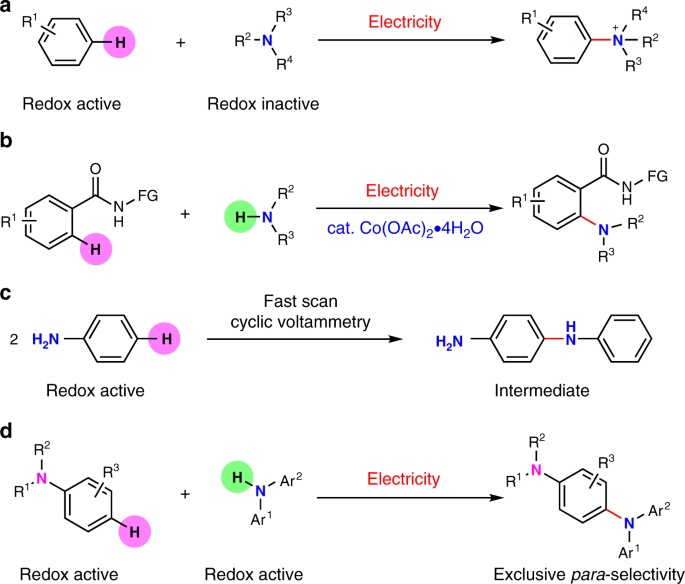
Electrooxidative para -selective C–H/N–H cross-coupling with hydrogen evolution to synthesize triarylamine derivatives | Nature Communications
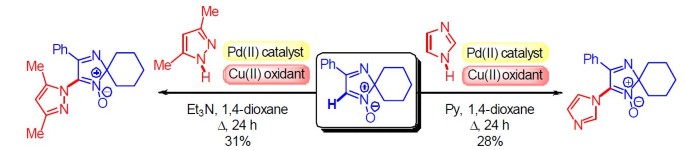
The First Example of Palladium(II)-Catalyzed Oxidative C–N Cross Coupling of 2 H -Imidazole 1-Oxide with Azoles | SpringerLink
![Molecules | Free Full-Text | Pharmacological Potential and Synthetic Approaches of Imidazo[4,5-b]pyridine and Imidazo[4,5-c]pyridine Derivatives | HTML Molecules | Free Full-Text | Pharmacological Potential and Synthetic Approaches of Imidazo[4,5-b]pyridine and Imidazo[4,5-c]pyridine Derivatives | HTML](https://www.mdpi.com/molecules/molecules-22-00399/article_deploy/html/images/molecules-22-00399-sch021.png)
Molecules | Free Full-Text | Pharmacological Potential and Synthetic Approaches of Imidazo[4,5-b]pyridine and Imidazo[4,5-c]pyridine Derivatives | HTML
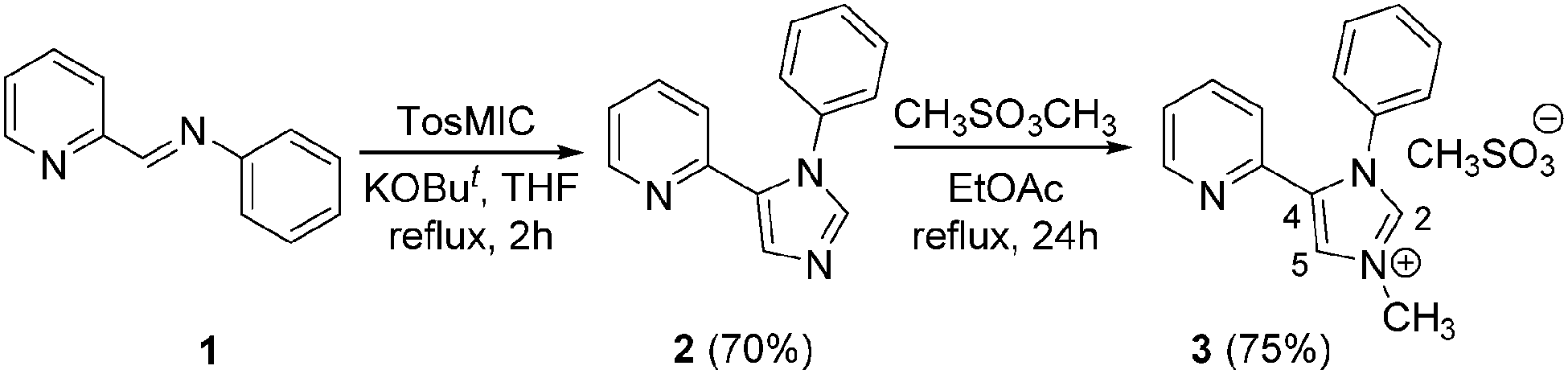
Chelation assistance as a tool for the selective preparation of an imidazole-based mesoionic palladium carbene complex - Chemical Communications (RSC Publishing) DOI:10.1039/C4CC07732F


![Bis(1 methyl 1H imidazole κN3)[N,N′ o phenylenebis(pyridine 2 carboxamido) κ4N]manganese(II) Bis(1 methyl 1H imidazole κN3)[N,N′ o phenylenebis(pyridine 2 carboxamido) κ4N]manganese(II)](https://data01.123dok.com/thumb/methyl-imidazole-phenyl-enebis-pyridine-carbox-amido-manganese-z3dx218y.Pki4ZiPT5VIqTD1so.jpeg)