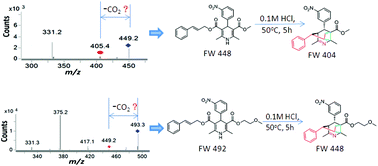
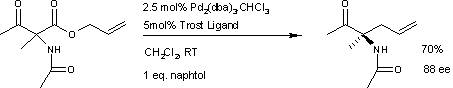Copper(II)- and palladium(II)-catalyzed enantioselective Claisen rearrangement of allyloxy- and propargyloxy-indoles to quaternary oxindoles and spirocyclic lactones. - Abstract - Europe PMC
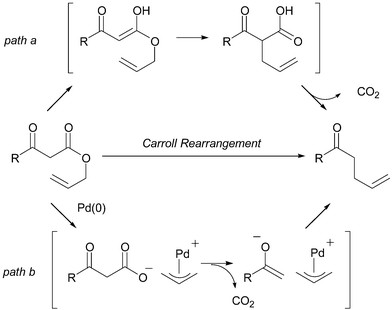
Asymmetric Carroll rearrangement of allyl α-acetamido-β-ketocarboxylates catalysed by a chiral palladium complex - Chemical Communications (RSC Publishing) DOI:10.1039/B505105C

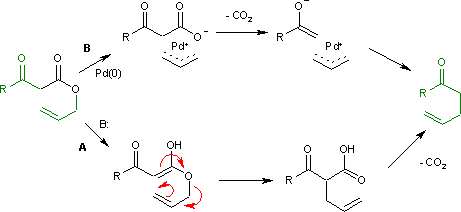
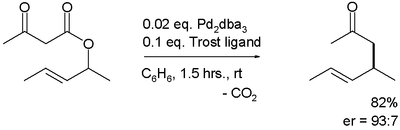
Palladium-catalyzed rearrangement of allylic esters of acetoacetic acid to give γ,δ-unsaturated methyl ketones

Enantioselective CpRu‐Catalyzed Carroll Rearrangement – Ligand and Metal Source Importance - Linder - 2008 - European Journal of Organic Chemistry - Wiley Online Library
Truong N. Nguyen The May lab Sigmatropic Rearrangements 1. Introduction - A class of concerted pericyclic reactions governed by

Palladium/N-heterocyclic carbene catalysed regio and diastereoselective reaction of ketones with allyl reagents via inner-sphere mechanism | Nature Communications

Introduction to the Development of a Catalytic Asymmetric Synthesis of Tertiary α-Aryl Ketones | SpringerLink
![Hiersemann M., Nubbemeyer U. (ed.) The Claisen Rearrangement. Methods and Applications [PDF] - Все для студента Hiersemann M., Nubbemeyer U. (ed.) The Claisen Rearrangement. Methods and Applications [PDF] - Все для студента](https://cv01.twirpx.net/0519/0519194.jpg?t=20181113225001)
Hiersemann M., Nubbemeyer U. (ed.) The Claisen Rearrangement. Methods and Applications [PDF] - Все для студента
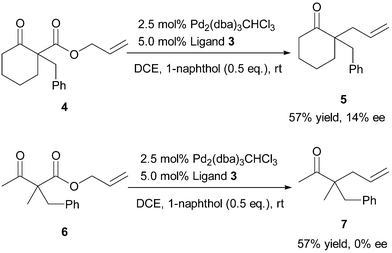
Asymmetric Carroll rearrangement of allyl α-acetamido-β-ketocarboxylates catalysed by a chiral palladium complex - Chemical Communications (RSC Publishing) DOI:10.1039/B505105C
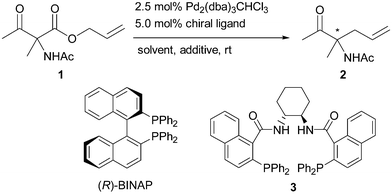
Asymmetric Carroll rearrangement of allyl α-acetamido-β-ketocarboxylates catalysed by a chiral palladium complex - Chemical Communications (RSC Publishing) DOI:10.1039/B505105C