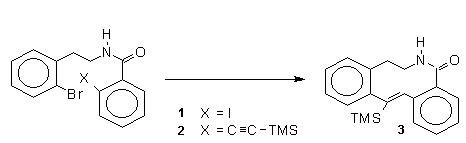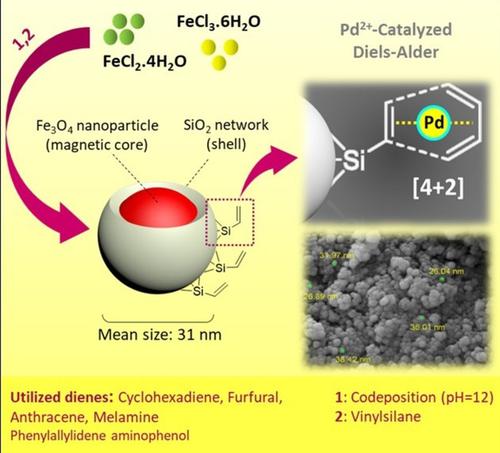
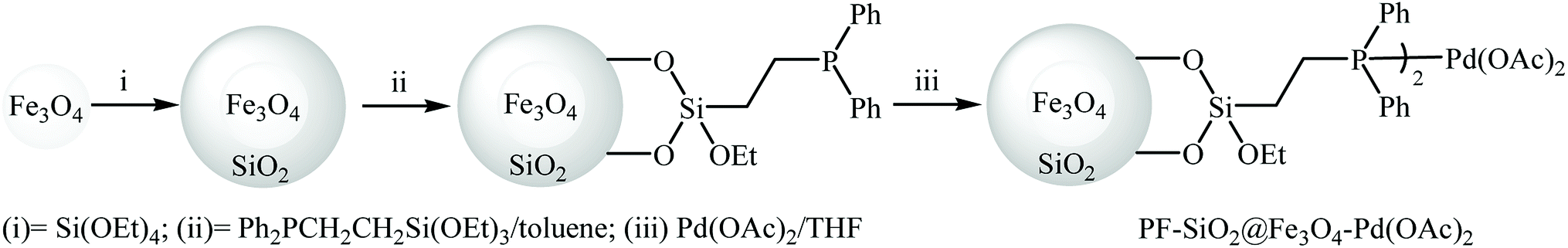
Surface functionalization of magnetic nanoparticles via palladium‐catalyzed Diels‐Alder approach - ChemistrySelect - X-MOL
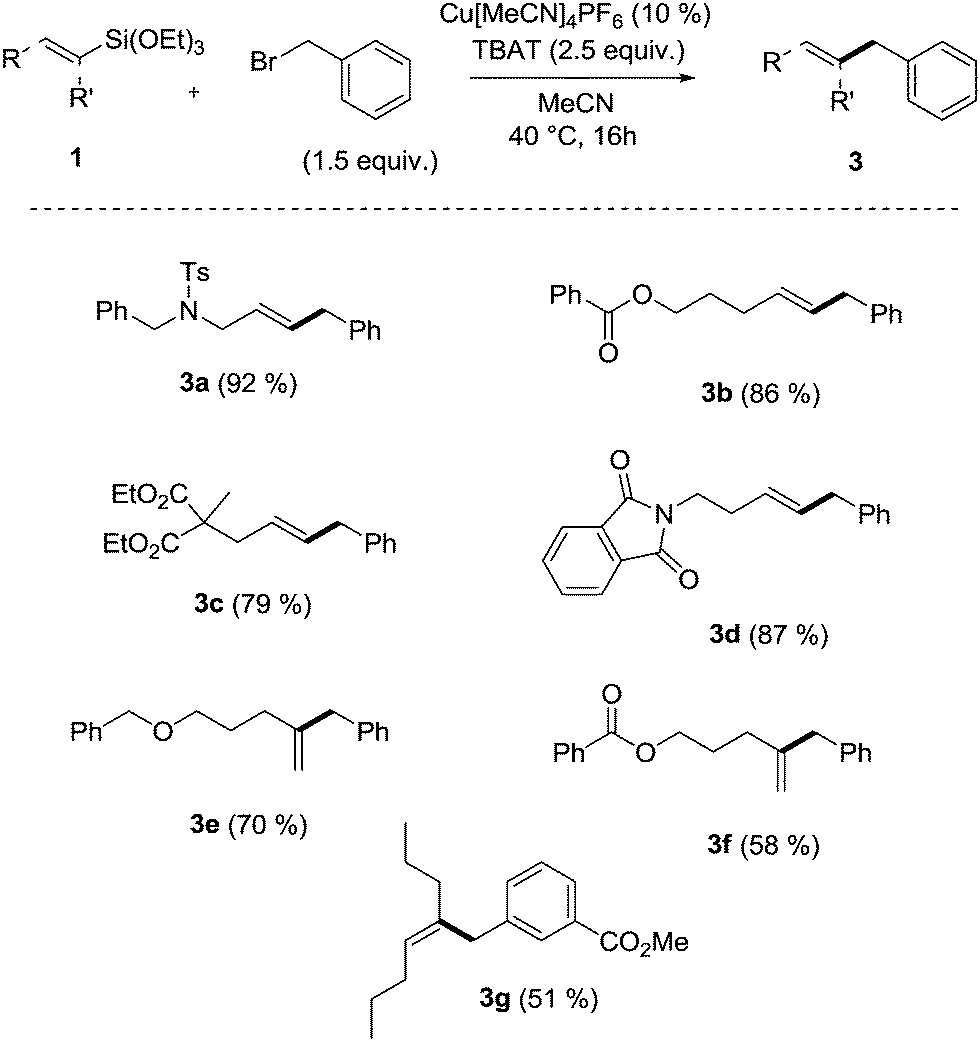
Copper-catalyzed Hiyama cross-coupling using vinylsilanes and benzylic electrophiles - Chemical Communications (RSC Publishing) DOI:10.1039/C4CC02923B
PdCl2(CH3CN)2-catalyzed regioselective C–H olefinations of 2-amino biaryls with vinylsilanes as unactivated alkenes - Chemical Communications (RSC Publishing)
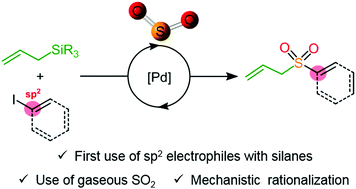
SO2 conversion to sulfones: development and mechanistic insights of a sulfonylative Hiyama cross-coupling - Chemical Communications (RSC Publishing)

Synthetic Applications of Allylsilanes and Vinylsilanes - Luh - - Major Reference Works - Wiley Online Library
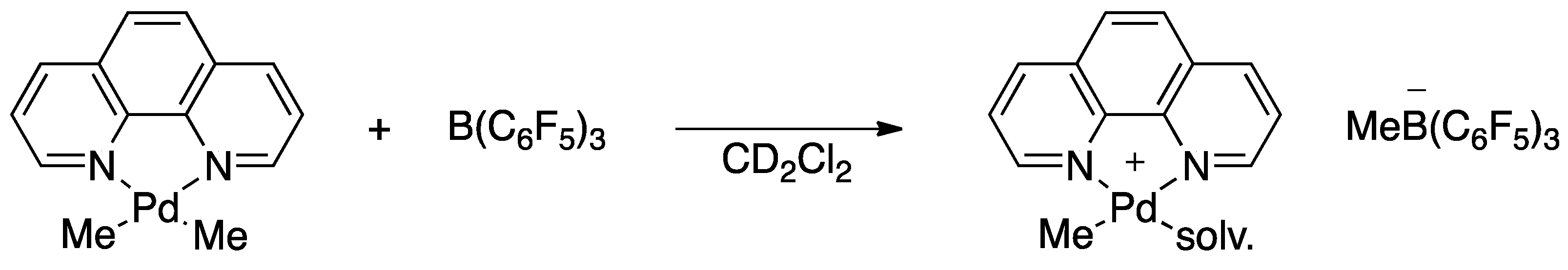
Inorganics | Free Full-Text | Non-Selective Dimerization of Vinyl Silanes by the Putative (Phenanthroline)PdMe Cation to 1,4-Bis(trialkoxysilyl)butenes | HTML
![PDF] Why does fluoride anion accelerate transmetalation between vinylsilane and palladium(II)-vinyl complex? Theoretical study. | Semantic Scholar PDF] Why does fluoride anion accelerate transmetalation between vinylsilane and palladium(II)-vinyl complex? Theoretical study. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c3f40d9baf4a5bdf783db2a5cc1200e5f5852ad3/3-Figure1-1.png)
PDF] Why does fluoride anion accelerate transmetalation between vinylsilane and palladium(II)-vinyl complex? Theoretical study. | Semantic Scholar

PDF) The effect of added silver nitrate on the palladium-catalyzed arylation of allyltrimethylsilanes

PDF) Non-Selective Dimerization of Vinyl Silanes by the Putative (Phenanthroline)PdMe Cation to 1,4-Bis(trialkoxysilyl)butenes
Recent advances in the application of nano-catalysts for Hiyama cross-coupling reactions - RSC Advances (RSC Publishing)

Table 1 from Palladium-Catalyzed Ortho-Silylation of Aryl Iodides with Concomitant Arylsilylation of Oxanorbornadiene: Accessing Functionalized ( Z)-β-Substituted Vinylsilanes and Their Analogues. | Semantic Scholar

Synthetic Applications of Allylsilanes and Vinylsilanes - Luh - - Major Reference Works - Wiley Online Library

Solvent interception, heterocyclization and desilylation upon NBS-induced sulfamidation of trimethyl(vinyl)silane. - Org. Biomol. Chem. - X-MOL
Palladium-catalyzed sequential three-component reactions to access vinylsilanes - Chemical Communications (RSC Publishing) DOI:10.1039/C8CC05254A

Preparation of Allyl and Vinyl Silanes by the Palladium‐Catalyzed Silylation of Terminal Olefins: A Silyl‐Heck Reaction - McAtee - 2012 - Angewandte Chemie - Wiley Online Library
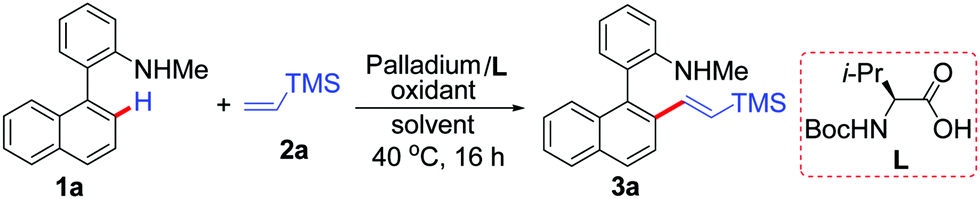
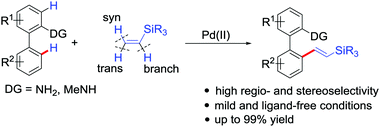
PdCl2(CH3CN)2-catalyzed regioselective C–H olefinations of 2-amino biaryls with vinylsilanes as unactivated alkenes - Chemical Communications (RSC Publishing)

Stereoselective Synthesis of Cis- and Trans-Tetrasubstituted Vinyl Silanes Using a Silyl-Heck Strategy and Hiyama Conditions for Their Cross-Coupling. - J. Am. Chem. Soc. - X-MOL

A Bithiophene‐Promoted ppm Levels of Palladium‐Catalyzed Regioselective Hydrosilylation of Terminal Allenes - Chen - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library
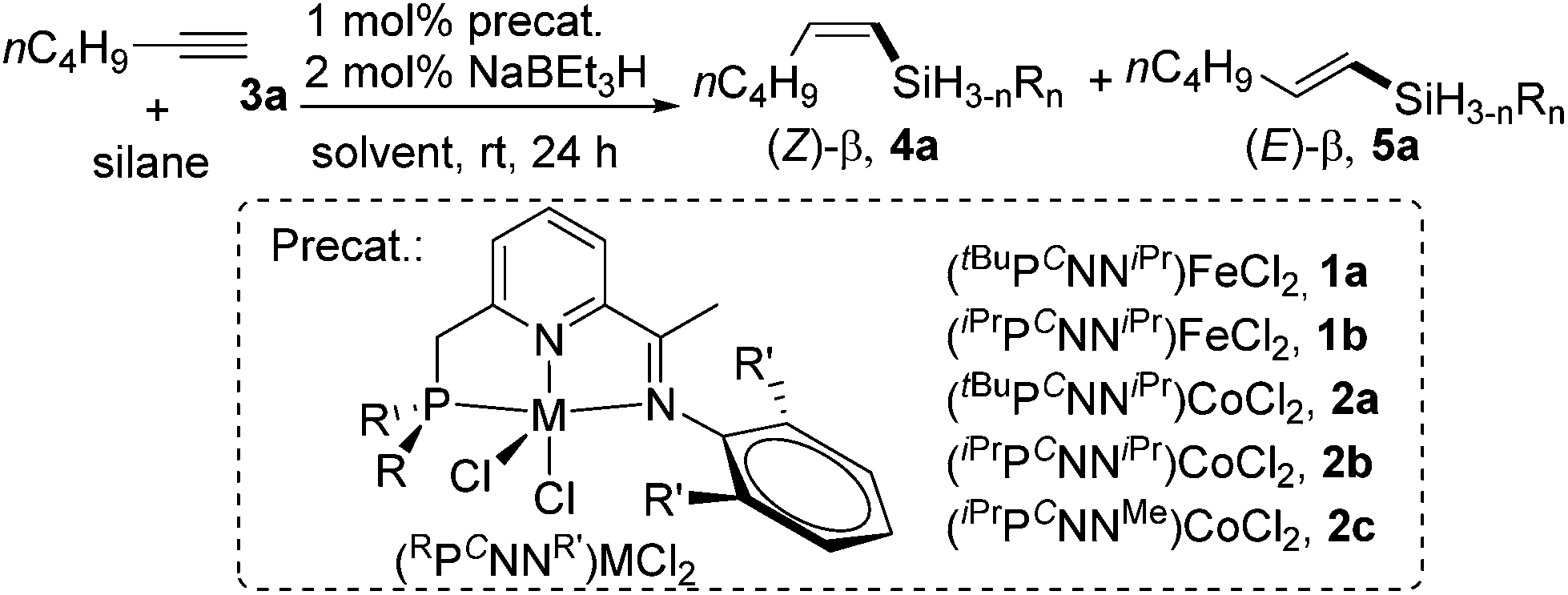
Pincer cobalt complex-catalyzed Z -selective hydrosilylation of terminal alkynes - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C7QO00250E
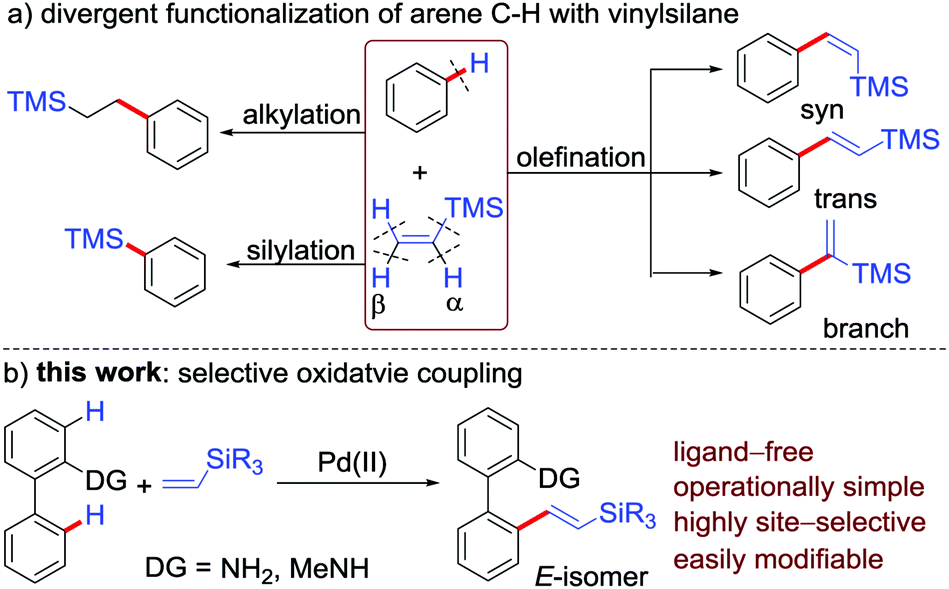
PdCl2(CH3CN)2-catalyzed regioselective C–H olefinations of 2-amino biaryls with vinylsilanes as unactivated alkenes - Chemical Communications (RSC Publishing)
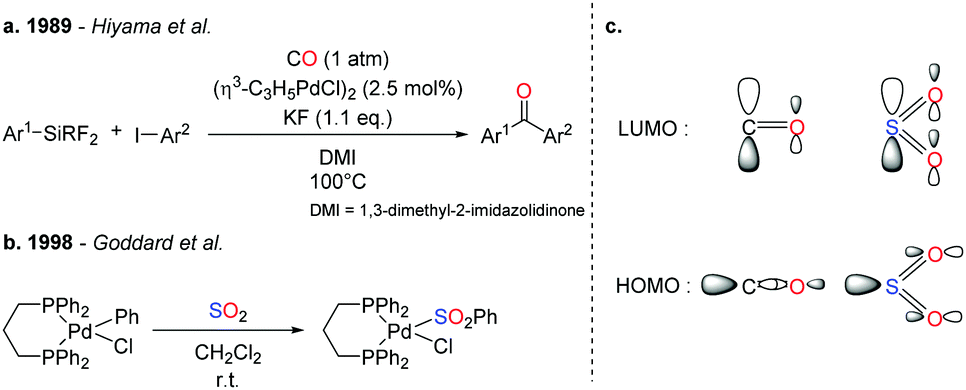
SO 2 conversion to sulfones: development and mechanistic insights of a sulfonylative Hiyama cross-coupling - Chemical Communications (RSC Publishing) DOI:10.1039/C9CC06858A

Supported palladium nanoparticles as heterogeneous ligand-free catalysts for the Hiyama C–C coupling of vinylsilanes and halobenzenes leading to styrenes - ScienceDirect